Blockers
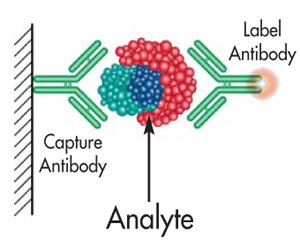
The existence of heterophilc antibodies and their potential for causing interference in immunoassays has been known for many years. The potentially devastating effects of false positive assay results on the patient and the medical community have more recently been delineated. The increasing use of the susceptible 2-site immunometric (“sandwich”) assay format has led to growing concern over the problem. For this reason, Scantibodies Laboratory has developed a unique heterophilic blocking reagent (HBR) product line that minimizes the occurrence of heterophilic antibody interference.
HETEROPHILIC BLOCKING REAGENT
![]() Qualification Test for Application of HBR
Qualification Test for Application of HBR
![]() Heterophilic Antibody Interference – What is it?
Heterophilic Antibody Interference – What is it?
![]() The Unique Aspects of HBR Over Conventional Blocking Methods
The Unique Aspects of HBR Over Conventional Blocking Methods
![]() Heterophilic Interference – What is Known?
Heterophilic Interference – What is Known?
![]() Heterophilic Antibodies – Sources
Heterophilic Antibodies – Sources
![]() Heterophilic Interference – How is it Detected?
Heterophilic Interference – How is it Detected?
![]() What is the Difference between “Heterophilic Antibody” and “HAMA?”
What is the Difference between “Heterophilic Antibody” and “HAMA?”
![]() What is a Blocking Reagent?
What is a Blocking Reagent?
![]() Characteristics of an Ideal Heterophilic Blocking Reagent
Characteristics of an Ideal Heterophilic Blocking Reagent
![]() The Scantibodies Heterophilic Blocking Reagent (HBR)
The Scantibodies Heterophilic Blocking Reagent (HBR)
![]() Performance Characteristics
Performance Characteristics
![]() False Positive Identification
False Positive Identification
![]() Certification
Certification
![]() Scantibodies Blocking Reagents for Assay Manufacturers
Scantibodies Blocking Reagents for Assay Manufacturers
![]() Scantibodies Blocking Reagents for Clinical Labs, Hospitals, etc.
Scantibodies Blocking Reagents for Clinical Labs, Hospitals, etc.
Product Information
SCANTIBODIES PRODUCTS FOR REDUCTION OF FALSE POSITIVES DURING ASSAY DEVELOPMENT
3KG775 – Assay Development Blocking Kit => Directional Insert | SDS
This Kit is provided as an aid in developing a reagent blocking formulation to eliminate false positive or negative interferences. Components that are included in this kit are useful for both active and passive blocking. The components in this kit have been selected to provide an array of options from which a selection may be made to develop a reagent blocking formulation.
SCANTIBODIES BLOCKING REAGENTS FOR ASSAY MANUFACTURERS (ACTIVE BLOCKERS)
3KC533 – Heterophilic Blocking Reagent (HBR), 20 mg/ml => Directional Insert | SDS
3KC533 contains specific murine immunoglobulins that block the heterophilic interaction by active binding to the heterophilic antibodies, which are capable of cross-linking the capture and the detection antibodies used in the immunoassay, resulting in false positive readings. The attachment of HBR-1 to the heterophilic antibodies blocks this cross-linking, and eliminates the interference caused by the heterophilic antibodies in the humoral fluids. In addition to its active blocking, this product is also characteristic of its passive blockage of the heterophilic interaction as well. The HBR is a liquid reagent with a protein concentration of 20 ± 2 mg/ml. The immunoglobulins are dissolved in a phosphate buffer with a pH of 7.2-7.6. The immunoglobulins in this product are at a purity of greater than or equal to 95%.
3KC576 – HBR-3 , Purified => SDS
Each vial contains approximately 4 mg of murine immunoglobulins. This product represents a variation in formulation with similar essential characteristics compared to HBR-1-Purified. The special formulation is designed to enhance its blocking capability at a lower concentration of immunoglobulins.
3KC542 – HBR-6 , Purified => SDS
Each vial of HBR-6 contains approximately 10 mg of immunoglobulins. This HBR-6 is specially formulated to enhance its heterophilic blocking ability. The immunoglobulins in this reagent are at a purity of greater than or equal to 95%.
3KC564 – HBR-9 , Purified => SDS
The HBR-9 is also one of our newly formulated products developed as an alternative for the HBR Plus, 3KC545. It contains murine immunoglobulins with different characteristics. It is specially formulated for application for immunoassays in which both the capture and detection antibodies are of murine origin. Like HBR Plus, this product is characteristic for its active as well as passive blocking efficacy. Each vial of this product contains approximately 20 mg of immunoglobulins, which are at a purity of greater than 90%, as shown by SDS-PAGE.
3KC565 – HBR-11 , Purified => SDS
3KC565 has been formulated with murine immunoglobulins. In addition to the products listed above, HBR-11 provides our customers with more selection for heterophilic blockage. The immunoglobulins in this product are at a purity of greater than or equal to 90%. It has a concentration of 10.0 +/- 1.0 mg/ml.
3KC545 – HBR-Plus , Purified => SDS
3KC545 was developed as an alternative for HBR-1. This product is compounded with immunoglobulins with different characteristics. Therefore, in addition to its active blocking characteristics, the special formulation and production procedures enhance its efficacy in its passive blocking ability as well. It has a concentration of 10.0 +/- 1.0 mg/ml.
3KC002 – HBR-21 => SDS
The HBR-21 is also one of our newly formulated products.. In addition to the products listed above, the HBR-21 provides our customers with more selection for heterophilic blockage. Each vial of this product contains approximately 10 mg of immunoglobulins.
3KC003 – HBR-22 => SDS
The HBR-22 is also one of our newly formulated products. In addition to the products listed above, the HBR-22 provides our customers with more selection for heterophilic blockage. Each vial of this product contains approximately 10 mg of immunoglobulins.
3KC006 – HBR-23 => SDS
The HBR-23 is also one of our newly formulated products. This product is formulated with murine immunoglobulins. In addition to the products listed above, the HBR-23 provides our customers with more selection for heterophilic blockage. Each vial of this product contains approximately 10 mg of immunoglobulins.
3KC007 – HBR-24 => SDS
The HBR-24 is also one of our newly formulated products. This product is formulated with murine immunoglobulins. In addition to the products listed above, the HBR-24 provides our customers with more selection for heterophilic blockage. Each vial of this product contains approximately 10 mg of immunoglobulins.
PASSIVE BLOCKERS
Mouse Immunoglobulin G (IgG) acts as a passive heterophilic blocker that neutralizes a subpopulation of the heterophilic antibody and reduces the heterophilic interactions. In addition, Mouse IgG also increases the free IgG concentration in an assay system and reduces the non-specific interaction that results in falsely elevated analyte concentrations. It is an inexpensive blocker that works well in a good number of assay formats. However, it does not have the active blocking ability as HBR does. Therefore, Mouse IgG may not be powerful enough to completely block certain strong heterophilic interactions.
3BM245 – Mouse lgG, Purified => SDS
This product is affinity-purified from normal mouse serum at a concentration of 10-12 mg/ml. It contains all the normal subclasses of mouse IgG, and is provided at a purity of greater than or equal to 95%, as determined by SDS-PAGE. Mouse IgG has known characteristics for passive blocking efficacy which makes it a good choice for a heterophilic blockage at a lower cost. Therefore, it is recommended for heterophilic blockage in immunoassays in which both the capture and detection antibodies are of murine origin.
3BM845 – Mouse lgG, Purified, Concentrated => SDS
This is a more concentrated (>35 mg/ml) version of our Mouse IgG.
3BG247 – Goat IgG, Liquid => SDS
It has a concentration of 10-30mg/ml, Buffer: 0.01M PBS, pH 7.4, 0.1% NaN3.
3BG568 – Goat IgG, Lyophilized => SDS
It has a concentration of 45-60mg/ml, Buffer: PBS(3mM Potassium Phosphate, 10mM Sodium Phosphate, 115mM Sodium Chloride, pH 7.2)no preservative added.
3BM203 – Mouse IgG, Purified => SDS
It has a concentration of 10-12mg/ml, Buffer: 0.1M Sodium Phosphate buffer, pH 7.4, 0.1% NaN3.
3BR230 – Rabbit IgG => SDS
It has a concentration of 5-15mg/ml, Buffer: 0.01M PBS, pH 7.4, no preservative added.
3BS251 – Sheep IgG => SDS
It has a concentration of 4.5 -5.5 mg/ml, Buffer 0.01M Phosphate buffer, 0.117M Sodium Chloride, pH 7.2, no preservative added.
SCANTIBODIES BLOCKING REAGENTS FOR CLINICAL LABS, HOSPITALS, ETC…
3IX761 – Nonspecific Antibody Blocking Tube (NABT) => Directional Insert
The NABT contains immunoglobulins. The non-specific antibodies in the serum or plasma samples bind to these immunoglobulins and are blocked from interfering in antibody detection immunoassays. Each tube contains enough reagent to inactivate the non-specific antibodies in 500 ?L of sample. The reagent is in the form of a lyophilized pellet at the bottom of the tube. The NABT allows for the rapid and simple elimination of false positive non-specific antibody interference in plasma or serum for detection assays (i.e., anti-HCV, HIV, Toxoplasmosis, Rubella, CMV. Herpes. Tg, TPO, etc.). The NABT is used as a sample treatment in preparation for testing. It can either be used in conjunction with the initial assay or in a secondary confirmation assay. NABT represents a sample pretreatment/second assay intended to confirm or disqualify the original FDA licensed non pretreatment assay result. The assay result from the pretreatment is NEVER TO BE USED AS A REPORTABLE RESULT. The pretreatment is only a confirmation aid designed to assist the lab to know whether to report the original non pretreatment assay result. In other words, if the pretreatment assay result is the same as the original assay result the original result is reported. However, if the pretreatment sample assay result is lower than the original result, the original result is not reported and the sample is submitted for further study for potential false positive assay interference.
3IX762 – Heterophilic Blocking Tubes (HBT) => Directional Insert
The HBT contains a unique blocking reagent composed of specific binders which inactivate heterophilic antibodies. Once the specific binders have bound to the heterophilic antibodies, the antibodies are no longer able to cause immunoassay interference. The reagent is in the form of a lyophilized pellet at the bottom of the tube. Each tube contains enough reagent to inactivate the heterophilic antibodies in 500 ?L of sample. The HBT allows for the rapid and simple elimination of false positive heterophilic interference in plasma or serum for immunoassays (i.e., FSH, LH, Prolactin, TSH, Ferritin, CEA, AFP, hCG, HBsAg, CK-MB, CA 1251, CA 19-9 , NSE, etc.). HBT represents a sample pretreatment/second assay intended to confirm or disqualify the original FDA licensed non pretreatment assay result. The assay result from the pretreatment is NEVER TO BE USED AS A REPORTABLE RESULT. The pretreatment is only a confirmation aid designed to assist the lab to know whether to report the original non pretreatment assay result. In other words, if the pretreatment assay result is the same as the original assay result the original result is reported. However, if the pretreatment sample assay result is lower than the original result, the original result is not reported and the sample is submitted for further study for potential false positive assay interference.